GelMA by Claro: great potential to build 3D models that mimic real tumors
The need for innovative 3D cancer models to enhance drug-testing process
The National Cancer Institute defines cancer as “a term for diseases in which abnormal cells divide without control and can invade nearby tissues. Cancer cells can also spread to other parts of the body through the blood and lymph systems.” [1]. Metastases are the primary cause of death from cancer. In 2018, cancer was the second leading cause of death worldwide, accounting for an estimated 9.6 million deaths. Lung, prostate, colorectal, stomach and liver cancer are common types of cancer in men, while breast, colorectal, lung, cervical and thyroid cancer are the most common among women [2].
Globally, researchers work hard to discover the best therapeutics and possible cure for cancer. The use of 2D cell cultures and animal models cannot predict efficiently the tumor response to anti-cancer drugs and frequently fail to identify the side-effects of drugs, or the interaction between cancer and immune system, because of the biologic imparity between human and animals. There is a need for innovative 3D cancer models to enhance drug-testing process with regard to the physiopathology of the tumor microenvironment [3]. A recent review published by Agustine et al. (2021) demonstrated that 3D bioprinting has a huge potential to build tissue models that can mimic tumor microenvironment at molecular, cellular, and physiological level [4].
Culturing cancer cells and modeling diseases with gelatin methacryloyl (GelMA)
Bioengineered 3D cancer models show potential to replace conventional screening tests and attend the demand of high-throughput automatized systems coupled with high-content imaging profiling [3]. 3D bioprinting is a fast-evolving biofabrication technique used to architect the microenvironment of tumor tissues. Cancer bioinks are comprised of patient-derived cancer cells and a biopolymer [4]. Many studies have demonstrated the suitability of gelatin methacryloyl (GelMA) for culturing cancer cells and modeling several types of diseases such as ovarian cancer metastases [5]. The methacryloyl groups attached on the gelatin backbone can be crosslinked by ultraviolet (UV)/vis light in presence of a photoinitiator. Irgacure 2959 or lithium phenyl‐2,4,6‐trimethylbenzoylphosphinate (LAP) are examples of cytocompatible photoinitiators. The stiffness of GelMA hydrogels can be controlled by the exposure time, the degree of substitution and photoinitiator concentration. Besides, GelMA hydrogels are transparent, allowing an easy microscopic visualization of cells [6].
The ideal GelMA for culturing cancer cells and modelling diseases
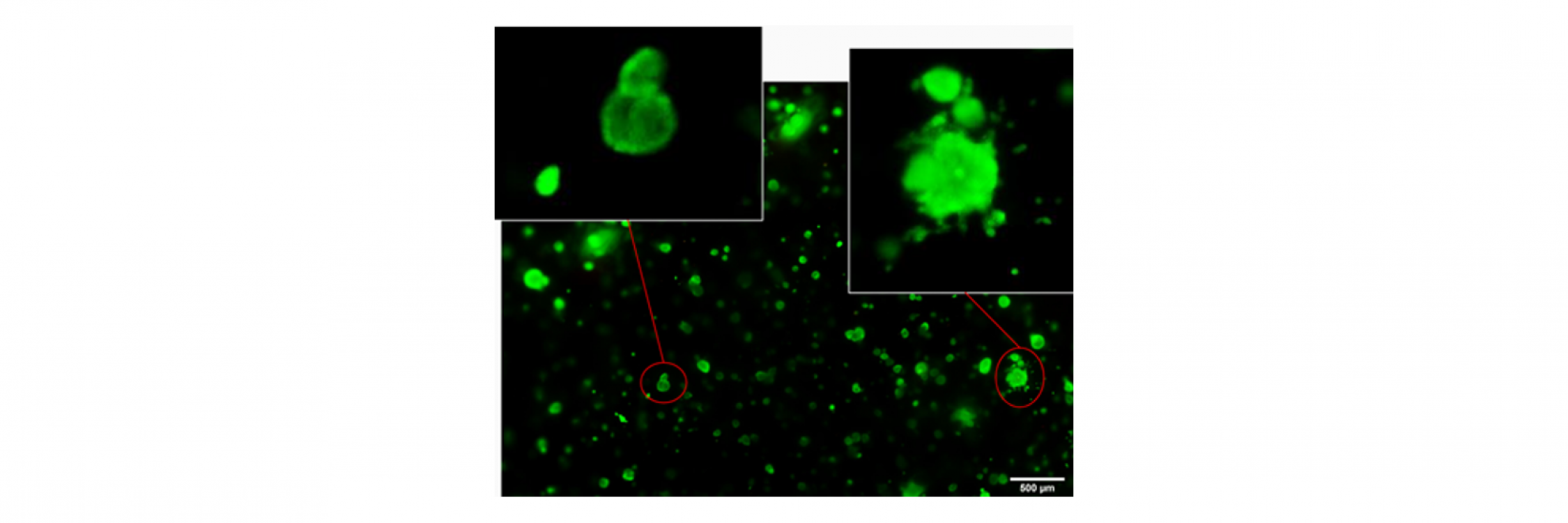
CLARO, part of Tessenderlo Group, offers the ideal gelatin methacryol (GelMA) for culturing tumor cells. A study collaboration between Claro and Allevi by 3D Systems showed that the stable and flexible photocrosslinked hydrogel made of ClaroBGI800 promoted cell viability higher than 80% throughout a LNCaP prostate cancer cells culture and evidence of tumor cell invasion [7] into the surrounding gelatin. The study highlights opportunities of personalizing tumor models using ClaroBGI800 hydrogel to support the proliferation of cells and biological materials.
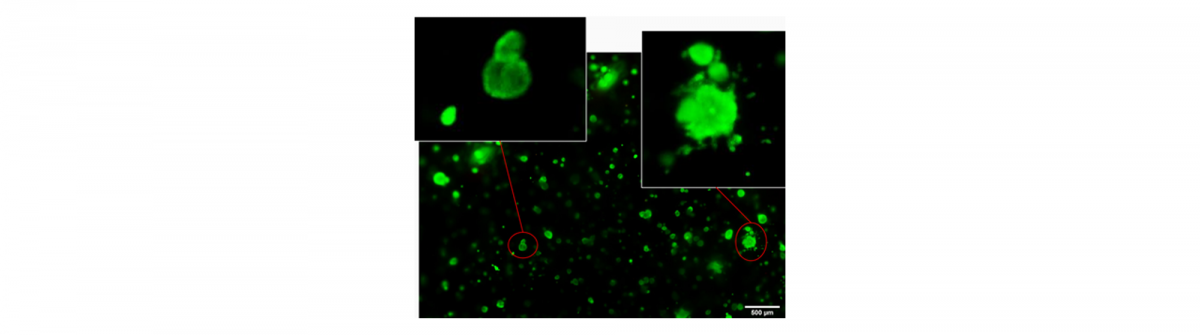
Picture taken at day 14 of culture by Allevi by 3D Systems using ClaroBGI800 hydrogel at concentration 10% w/v. LNCaP prostate cancer cells were cultured in T75 flasks at 37°C, 5% CO2, 85% humidity. 20,000 LNCaP cells were seeded per 100ul gel. 5 seconds of photocrosslinking at ~10mW/cm2.
The stiffness of the construct can be further optimized by controlling the bioink composition, process of biofabrication and maturation conditions. Post-maturation, personalized 3D tumor models can be exploited to test an range of potential chemotherapeutic agents. The effects of chemotherapeutic agents on cell viability and tumor growth reduction can be carried out in multi-well plates, producing large numbers of homogeneous organoids therefore improving the throughput of screening [4].
References
[1] NIH, “Cancer - NCI Dictionaries.” https://www.cancer.gov/publications/dictionaries/cancer-terms/def/cancer (accessed May 17, 2021).
[2] WHO, “Cancer,” 2021. https://www.who.int/health-topics/cancer#tab=tab_1 (accessed May 17, 2021).
[3] V. Brancato, J. M. Oliveira, V. M. Correlo, R. L. Reis, and S. C. Kundu, “Could 3D models of cancer enhance drug screening?,” Biomaterials, vol. 232, p. 119744, 2020, doi: https://doi.org/10.1016/j.biomaterials.2019.119744.
[4] R. Augustine et al., “3D Bioprinted cancer models: Revolutionizing personalized cancer therapy,” Transl. Oncol., vol. 14, no. 4, p. 101015, 2021, doi: https://doi.org/10.1016/j.tranon.2021.101015.
[5] K. Shield, M. L. Ackland, N. Ahmed, and G. E. Rice, “Multicellular spheroids in ovarian cancer metastases: Biology and pathology,” Gynecol. Oncol., vol. 113, no. 1, pp. 143–148, 2009, doi: https://doi.org/10.1016/j.ygyno.2008.11.032.
[6] B. Blanco-Fernandez, V. M. Gaspar, E. Engel, and J. F. Mano, “Proteinaceous Hydrogels for Bioengineering Advanced 3D Tumor Models,” Adv. Sci., vol. 8, no. 4, 2021, doi: 10.1002/advs.202003129.
[7] E. B. Berens, J. M. Holy, A. T. Riegel, and A. Wellstein, “A Cancer Cell Spheroid Assay to Assess Invasion in a 3D Setting,” J. Vis. Exp., no. 105, p. 53409, Nov. 2015, doi: 10.3791/53409.